
Background
Research indicates that 2′-fucosyllactose (2′-FL) specifically stimulates the proliferation of Bifidobacterium species, thereby establishing a healthy gut microbial environment. Escherichia coli O157 is a common pathogen capable of causing severe haemorrhagic colitis and haemolytic uraemic syndrome (HUS), particularly in children, the elderly, and immunocompromised individuals. Given its potent virulence and high antibiotic resistance, identifying effective preventive and therapeutic strategies is paramount. Against this backdrop, 2′-FL, as a functional food ingredient, may play a significant role in preventing and mitigating E. coli O157 infections.
Study Design and Methods
This study employed a mouse infection model to analyse the inhibitory effect of 2′-FL on intestinal E. coli O157 infection following dietary administration. Experiments included extracting bacterial DNA from mouse caecal contents, performing PCR amplification and gene analysis, and utilising 16S rDNA gene sequencing to analyse the gut microbiome. Inflammation levels were assessed by measuring IL-6, TNF-α, and IL-1β concentrations in blood, ileum, and colon. Additionally, qPCR methods were employed to detect expression levels of mucin-2 (MUC2) and tight junction protein-related genes.
Key Findings
1. 2′-FL inhibits Escherichia coli O157 colonisation in the gut
In both the ileum and colon, the colonisation rate of Escherichia coli O157 in the 2′-FL group was significantly lower than that in the control group (MC group), at 8.63% ± 6.87% and 6.16% ± 2.25% respectively. This indicates that 2′-FL possesses a significant anti-pathogenic colonisation effect, contributing to a reduction in the intestinal pathogen burden.
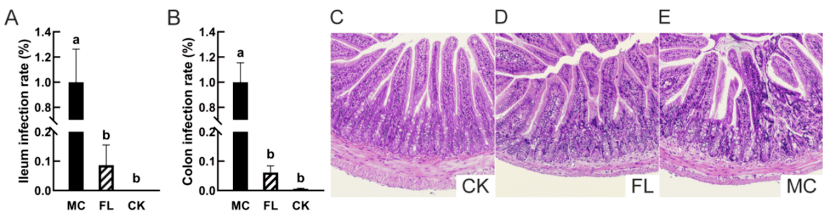
Figure 1: Colonisation of Escherichia coli O157 in the ileum and pathological sections of ileal tissue. (A) Colonisation in the ileum; (B) Colonisation in the colon; (C) Representative image of ileal tissue section from the control group (CK) (HE staining, ×100); (D) Representative image of ileal tissue section from the FL group (HE staining, ×100); (E) Representative image of ileal tissue section from the model control group (MCG) (HE staining, ×100).
2. Effects on Gut Microbiota Composition and Community Structure
At the phylum level, prolonged supplementation with 2′-FL significantly reduced the abundance of Firmicutes and Patescibacteria, while increasing that of Bacteroidetes and Proteobacteria. Specifically, at the genus level, Parabacteroides, Eubacterium, Mucispirillum, Anaerotruncus, and Alistipes were markedly diminished. These alterations reflect 2′-FL's substantial influence on gut microbial ecological equilibrium. Furthermore, the 2′-FL group exhibited heightened diversity and stability. Such changes not only aid in resisting pathogenic colonisation but may also enhance overall host health by promoting the growth of beneficial bacteria.
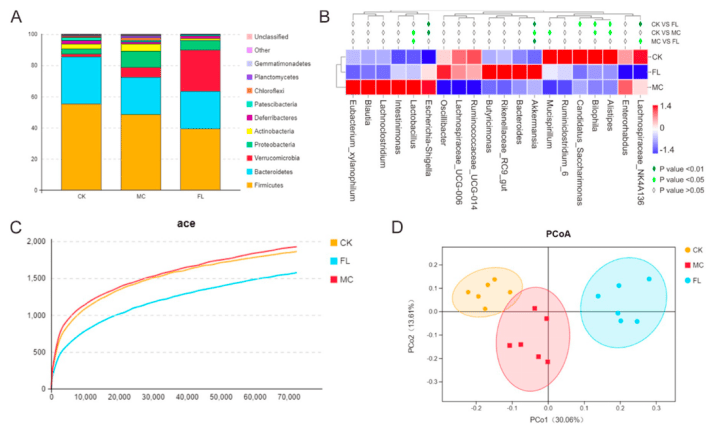
Figure 2: Relative abundance, diversity, and functional prediction of gut microbiota in different mouse groups: (A) Phylum; (B) Genus; (C) Dilution curve; (D) PCoA analysis based on operational taxonomic unit (OTU) results.
3. Effects on Inflammation Levels, Gene Expression, and Host Immune Responses 2′-FL significantly reduced levels of inflammatory cytokines (IL-6, TNF-α, and IL-1β) in the ileum and colon of mice, indicating its efficacy in suppressing gut inflammatory responses triggered by pathogenic bacterial infection. Concurrently, 2′-FL upregulated the expression of mucin-2 (MUC2) and tight junction protein genes in the colon. Enhanced expression of these genes contributes to strengthened intestinal barrier function, reducing pathogen invasion and colonisation. Moreover, following 2′-FL supplementation, the host immune system demonstrated improved regulatory capacity, manifested as a balanced profile of pro-inflammatory and anti-inflammatory cytokines. This immunomodulatory effect may represent an indirect action of 2′-FL mediated through the gut microbiota.
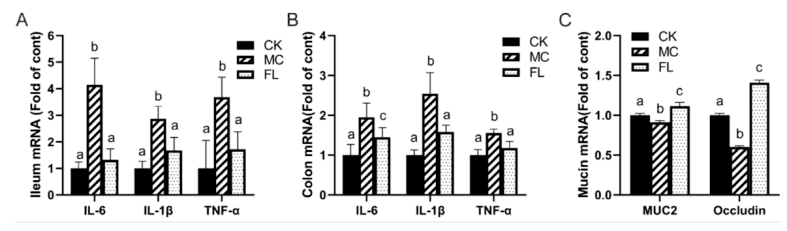
Figure 3: Relative mRNA levels of inflammatory cytokines and mucins, normalised against β-actin mRNA expression: (A) inflammatory cytokines in the ileum; (B) inflammatory cytokines in the colon; (C) mucin expression in the colon. Different letters (a–c) denote significant differences in the same factor between groups (p < 0.05).
Discussion and Outlook
Research indicates that 2′-FL exerts significant effects in regulating the gut microbiota and reducing pathogenic bacterial colonisation, particularly demonstrating inhibitory activity against Escherichia coli O157. Furthermore, its efficacy in ameliorating intestinal inflammation and enhancing barrier function has been validated. Nevertheless, further investigations are required to elucidate its precise mechanisms and explore its potential applications against other pathogenic bacterial infections. Future investigations may elucidate its comprehensive role in gut health by exploring synergistic interactions between 2′-FL and other prebiotics and probiotics.
Reference
[1] Wang Y , Zou Y , Wang J ,et al.The Protective Effects of 2′-Fucosyllactose against E. Coli O157 Infection Are Mediated by the Regulation of Gut Microbiota and the Inhibition of Pathogen Adhesion[J].Nutrients, 2020, 12(5):1284.DOI:10.3390/nu12051284.
