
Background
Human milk oligosaccharides (HMOs) play a pivotal role in infant gut health and immune system development. Among these, 2′-fucosyllactose (2′-FL) is one of the most prevalent HMOs. 2′-FL not only promotes the growth of beneficial bacteria but also exhibits anti-infective and anti-inflammatory properties, particularly in regulating intestinal inflammatory responses. In intestinal inflammation, CD14 serves as a pivotal receptor that recognises and binds bacterial components such as lipopolysaccharide (LPS), thereby initiating immune responses. IL-8, a pro-inflammatory cytokine, attracts immune cells to the site of infection during the inflammatory process, amplifying the inflammatory response. 2′-FL can mitigate inflammation triggered by intestinal pathogens to some extent by regulating CD14 expression and IL-8 release.
Study Design and Methods
This study employed human intestinal epithelial cells to simulate the intestinal environment. Cells were first exposed to LPS to induce inflammation, followed by the addition of 2′-FL across different experimental groups to observe its effects on CD14 expression. Gene and protein expression levels of CD14 were assessed via quantitative PCR and Western blot analysis, while IL-8 release was evaluated using ELISA to determine 2′-FL's anti-inflammatory efficacy. Additionally, the study investigated whether 2′-FL influences pathogen adhesion capacity to intestinal epithelial cells.
Key Findings
1. Suppressed CD14 Expression
Following LPS treatment, CD14 expression in intestinal epithelial cells increased significantly, indicating successful induction of the inflammatory response. However, the cell group treated with 2'-FL exhibited markedly reduced levels of both CD14 gene and protein expression, demonstrating 2'-FL's role in mitigating the inflammatory response by downregulating CD14 expression.
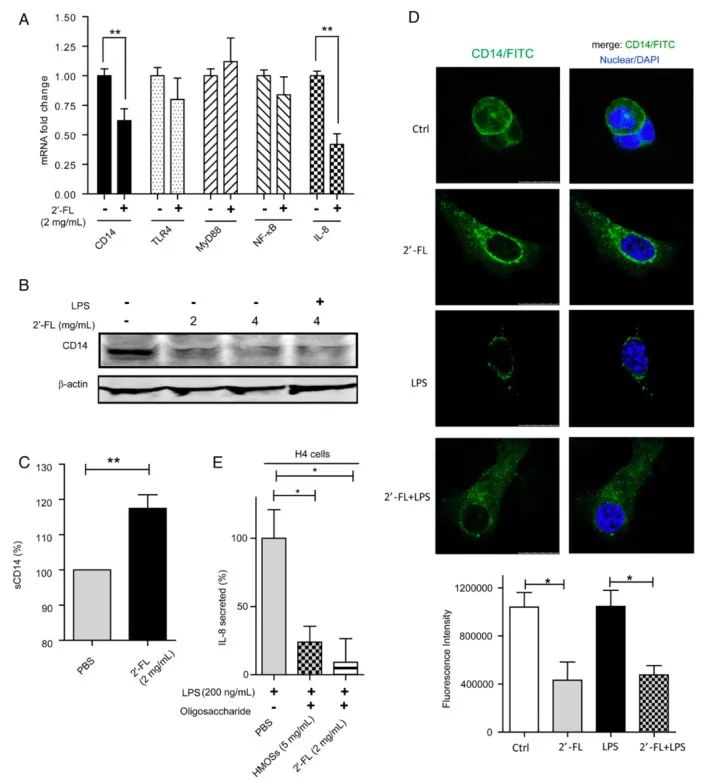
Figure 1: 2'-FL alters CD14 expression and distribution in cells
T84 cells treated with 2'-FL for 48 hours were subsequently challenged with 100 μg/mL LPS to measure 2'-FL-induced changes in CD14 mRNA levels, protein levels, and intracellular distribution.
2. Reduced IL-8 secretion
Following LPS treatment, IL-8 release from intestinal epithelial cells increased significantly, reflecting heightened inflammation. However, in the experimental group treated with 2′-FL, IL-8 release was markedly diminished, suggesting that 2′-FL may mitigate inflammatory responses by inhibiting CD14-mediated signalling pathways.
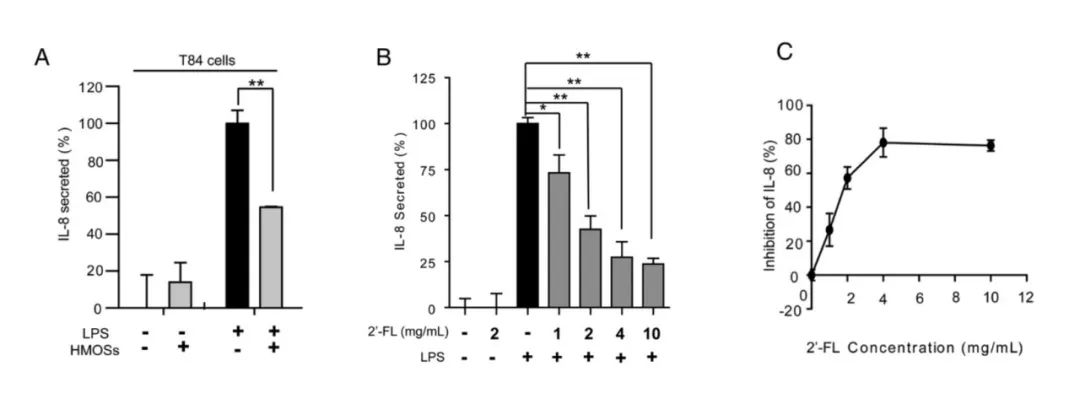
Figure 2: 2'-FL Inhibits LPS-Induced IL-8
Cells were treated with 2'-FL for 48 hours, then challenged with LPS. The degree of inflammatory response was measured by IL-8 levels.
3. Pathogen adhesion is inhibited
Research has also revealed that 2′-FL significantly reduces pathogen adhesion to intestinal epithelial cells. This may occur because 2′-FL mimics the sugar chains on the surface of intestinal epithelial cells, competitively preventing pathogens from binding to host cells and thereby lowering the risk of infection.
Discussion and Outlook
This study elucidates the potential mechanism by which 2′-FL modulates LPS-induced intestinal inflammatory responses, demonstrating that 2′-FL not only plays a crucial role in preventing infection but also alleviates inflammation triggered by intestinal pathogens by regulating immune responses. This holds potential benefits for intestinal health in both infants and adults. Future research may further validate the anti-inflammatory effects of 2′-FL in more complex in vivo models, particularly regarding its application in diseases associated with intestinal inflammation. Moreover, given the diverse array of HMOs present in human milk, the potential of other oligosaccharides in immunomodulation warrants exploration. The clinical application prospects for 2′-FL are extensive, extending beyond infant formula to its potential use as a natural anti-inflammatory agent in adult inflammatory bowel disease (IBD) and other gut disorders. Consequently, future research should prioritise establishing the safety and efficacy of 2′-FL, thereby providing scientific justification for its incorporationfunctional foods or pharmaceutical development.
Reference
[1] He, YingYing, et al. “The human milk oligosaccharide 2′-fucosyllactose modulates CD14 expression in human enterocytes, thereby attenuating LPS-induced inflammation.” Gut 65.1 (2016): 33-46.
