
Background
Vaccination is pivotal in enhancing human resistance to infectious diseases. However, infants, young children, and other immunocompromised individuals often struggle to generate sufficient antibodies post-vaccination, thereby compromising the vaccine's intended protective efficacy. 2′-Fucosyllactose (2′-FL), a key oligosaccharide in human milk, possesses immune-enhancing properties and can bolster vaccine protection by stimulating the immune system.
Study Design and Methods
This study employed an influenza vaccination model. Six-week-old female mice were dividedgroups and administered dietary interventions with varying concentrations of 2′-FL (0.25%–5%) prior to vaccination. To evaluate 2′-FL's impact on immune function, assessments were conducted on delayed-type hypersensitivity (DTH), serum immunoglobulin (IgG1 and IgG2a) levels, B-cell activation, and T-cell proliferation. DTH represents a reaction occurring within days of vaccination, reflecting the body's defensive capacity against specific antigens. Higher serum immunoglobulin levels (IgG1 and IgG2a) typically indicate stronger humoral immunity. B-cell activation and T-cell proliferation, meanwhile, reflect the immune system's active response to the vaccine.
Key Findings
1. 2′-FL Enhances DTH Immune Response
Dietary 2′-FL significantly amplified the DTH response in mice, indicating markedly enhanced immune defence against specific antigens. The intensity of the DTH response correspondingly increased with rising 2′-FL concentrations, demonstrating that 2′-FL effectively promotes the immune system's defensive capabilities.
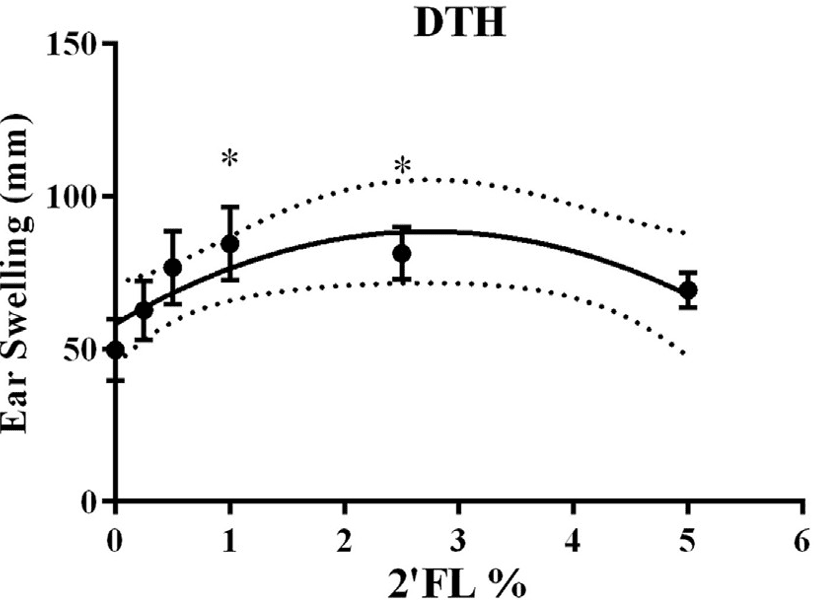
Figure 1: Effects of 2′-FL dietary intervention on vaccine-specific delayed-type hypersensitivity (DTH)
2. 2′-FL elevates immunoglobulin levels
Research indicates that serum levels of immunoglobulins IgG1 and IgG2a were significantly elevated in mice treated with 2′-FL. These immunoglobulins constitute vaccine-induced specific antibodies, and their increase signifies enhanced humoral immunity.
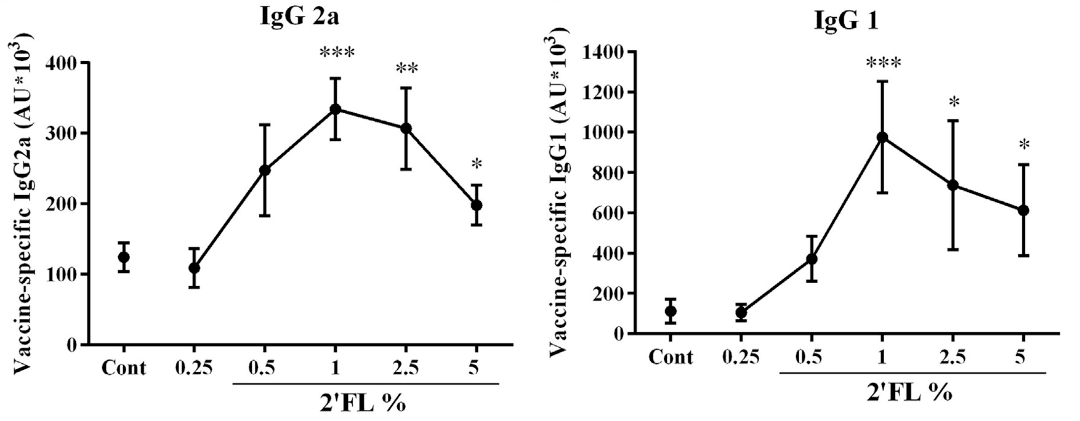
Figure 2: Effect of 2′-FL dietary intervention on antibody levels in serum collected on day 31
3. 2′-FL promotes B-cell activation
Flow cytometry analysis revealed significantly heightened levels of B-cell activation in the spleen and mesenteric lymph nodes of mice administered 2′-FL. This indicates enhanced antibody production capacity, facilitating more effective neutralisation of invading pathogens.
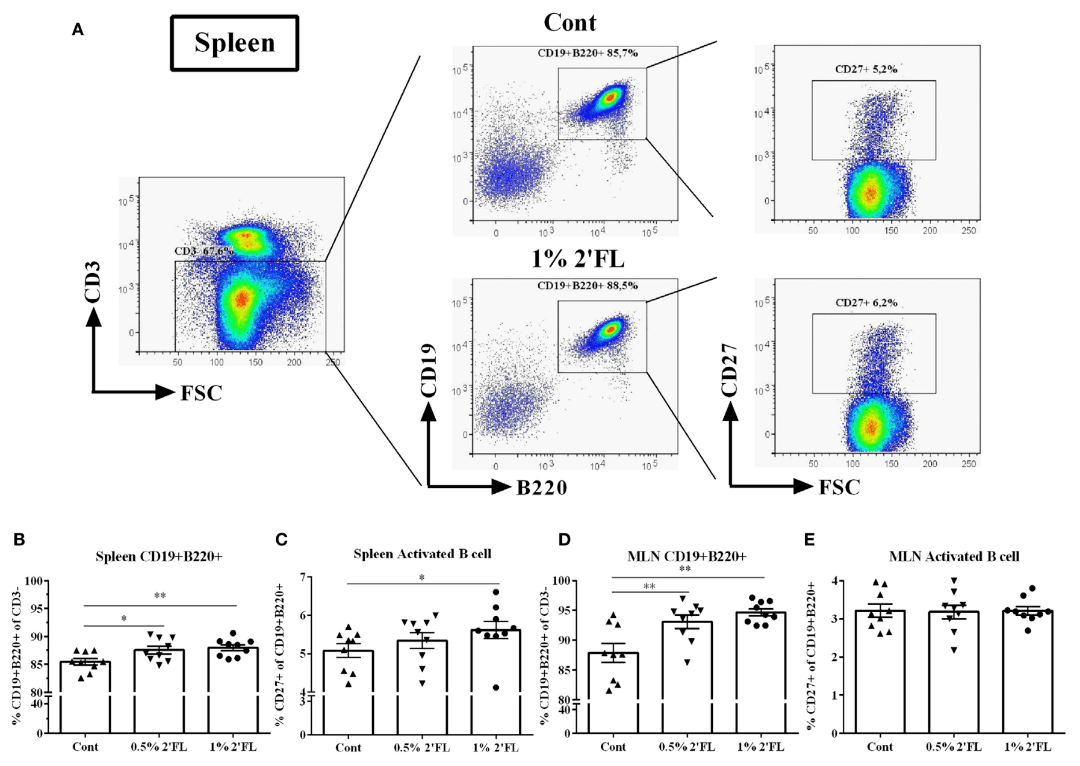
Figure 3: Flow cytometric analysis of B-cell subsets in spleen and mesenteric lymph nodes (MLN) collected on day 31 following 2′-FL dietary intervention.
4. 2′-FL Promotes T-Cell Proliferation
Research has demonstrated that 2′-FL significantly enhances the proliferation of both CD4+ and CD8+ T cells, thereby strengthening the protective function of vaccine-specific cellular immunity. The increase in CD4+ T cells aids in activating other immune cells, while CD8+ T cells are responsible for directly attacking infected cells. This indicates that 2′-FL plays a positive role in enhancing overall immune function.
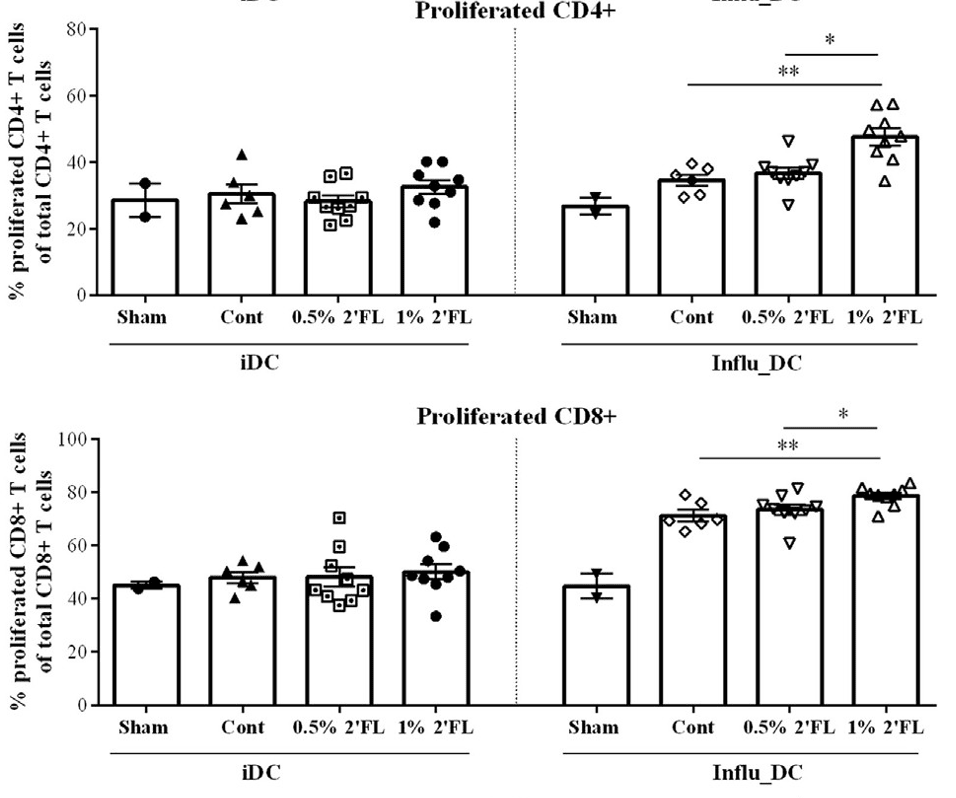
Figure 4: Percentage of CD4+ and CD8+ T cells proliferating following re-stimulation in vitro of bone marrow dendritic cells (BMDCs) loaded with influenza virus
Discussion and Outlook
This study demonstrates that 2′-FL exhibits significant efficacy in enhancing vaccine immunogenicity. This is manifested through enhanced DTH responses, markedly elevated serum immunoglobulin levels (IgG1 and IgG2a), increased B-cell activity, and T-cell proliferation. These findings indicate that 2′-FL effectively boosts both humoral and cellular immunity, providing a theoretical foundation for its potential application as an immunostimulant in infant formula. Future research may further explore the potential application of 2′-FL in human vaccination programmes and evaluate its long-term benefits in neonatal immune development, particularly its role in promoting immunological health.
Reference
[1] Xiao L, Leusink-Muis T, Kettelarij N, et al. Human milk oligosaccharide 2′-fucosyllactose improves innate and adaptive immunity in an influenza-specific murine vaccination model[J]. Frontiers in immunology, 2018, 9: 452.
