
Background
Skin inflammations such as psoriasis are caused by excessive proliferation of skin cells and persistent inflammatory responses triggered by immune system dysregulation. Research indicates that 2′-fucosyllactose (2′-FL), a key oligosaccharide in human milk, alleviates symptoms of skin inflammations like psoriasis by modulating relevant immune responses. This study explores the potential mechanisms by which 2′-FL inhibits psoriasis-like skin inflammation, offering novel insights for alleviating such conditions.
Study Design and Methods
A psoriasis mouse model induced by imiquimod (IMQ) was employed. Six-week-old female mice were dividedgroups receiving dietary interventions with varying concentrations of 2′-FL (0.25%–5%). Skin inflammation severity was assessed using the Psoriasis Area and Severity Index (PAS). To elucidate the specific regulatory mechanisms of 2′-FL on psoriasiform skin inflammation, the study analysed the proportion of Th17 cells—inflammatory cells associated with psoriasis—via flow cytometry and Western blot techniques. It further examined alterations in STAT3, a key signalling pathway regulating Th17 cells, and the expression of RORyt, a crucial transcription factor influencing Th17 cell differentiation.
Key Findings
Th17 cells and their associated pro-inflammatory cytokines exert a pivotal influence in the pathogenesis of psoriasis. 2′-FL significantly suppressed cutaneous inflammation by modulating STAT3—a key signalling pathway in immune response and inflammatory regulation—and RORyt, a crucial transcription factor governing Th17 cell differentiation.
2′-FL markedly ameliorates psoriasiform skin inflammation
Mice treated with 2′-FL exhibited significantly improved skin inflammation, with marked reductions in erythema, scaling, and skin thickness, alongside a significant decrease in PAS scores. This demonstrates 2′-FL's efficacy in alleviating cutaneous inflammatory responses.
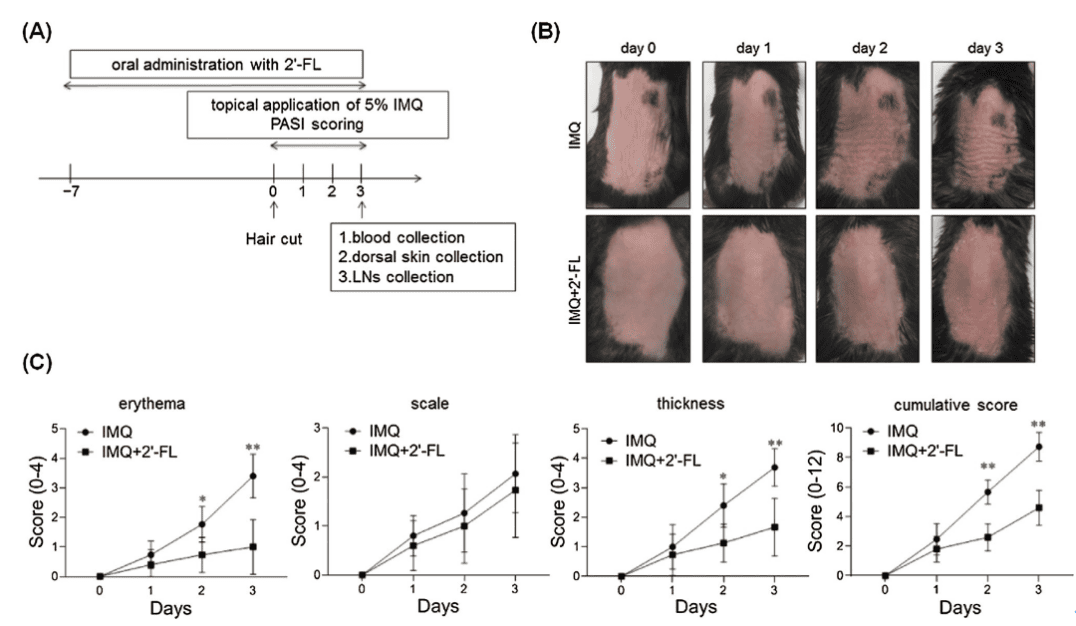
Figure 1: Mice treated with 2′-FL exhibit a reduced inflammatory response to IMQ
2′-FL effectively reduced skin inflammation by decreasing the expression of Th17 cells and their associated pro-inflammatory factors.
Further analysis revealed that the proportion of Th17 cells in mice treated with 2′-FL was significantly reduced, with a corresponding decrease in the expression of related pro-inflammatory factors such as IL-17 and IL-22, thereby effectively mitigating the skin inflammatory response.
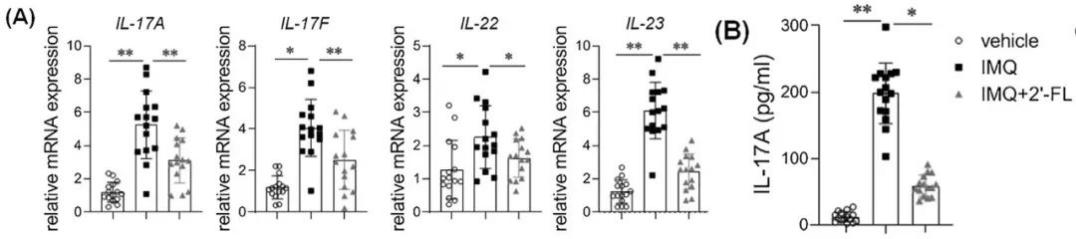
Figure 2: 2′-FL reduced the mRNA expression of Th17-associated cytokines in skin lesions of IMQ-treated mice.
2′-FL reduces the expression of Th17 cells and their associated pro-inflammatory cytokines by modulating the STAT3 signalling pathway and the cellular transcription factor RORyt.
Further research has elucidated the specific mechanism by which 2′-FL inhibits Th17 cells. By suppressing the phosphorylation of the STAT3 signalling pathway and the expression of the cellular transcription factor RORyt, 2′-FL effectively prevents the excessive differentiation and activation of Th17 cells, thereby inhibiting inflammatory responses.
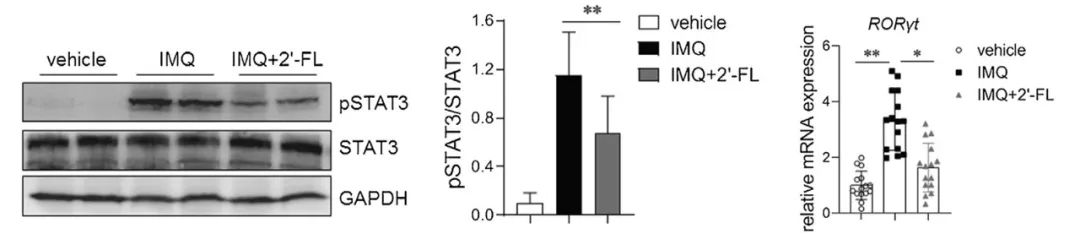
Figure 3: 2′-FL reduced STAT3 phosphorylation and the relative mRNA expression levels of RORγt in skin lesions of IMQ-treated mice.
Discussion and Outlook
This study demonstrates that 2′-FL exerts a significant inhibitory effect on cutaneous inflammation. By modulating STAT3—a key signalling pathway in immune responses and inflammatory regulation—and RORγt—a crucial transcription factor in Th17 cell differentiation—2′-FL reduces the expression of inflammatory Th17 cells and their associated pro-inflammatory mediators. This effectively suppresses the progression of psoriatic symptoms, offering novel therapeutic insights for psoriasis and other inflammatory skin conditions. Future research may further explore the application potential of 2′-FL in human immune-mediated skin disorders, particularly its role in the long-term management of chronic inflammatory and autoimmune conditions.
Reference
[1] Lei K, Wang D, Lin L, et al. 2′-fucosyllactose inhibits imiquimod-induced psoriasis in mice by regulating Th17 cell response via the STAT3 signaling pathway[J]. International Immunopharmacology, 2020, 85: 106659.
