
Background
Inflammatory bowel disease (IBD) is a chronic condition characterised by persistent intestinal inflammation, with colitis being one of its most prevalent manifestations. Research indicates that IBD development is frequently associated with gut microbiota dysbiosis and immune system hyperreactivity. 2′-Fucosyllactose (2′-FL), a key component of human milk oligosaccharides (HMOs), contributes to establishing a healthy gut microbiota and modulating immune responses. This study aims to evaluate the efficacy of 2′-FL in alleviating colitis and explore its potential for improving IBD-related symptoms.
Study Design and Methods
A DSS (d-starch sulphate) induced colitis mouse model was employed, with mice dividedcontrol, low-dose 2′-FL, high-dose 2′-FL, and other groups. The efficacy of 2′-FL in alleviating IBD-related symptoms was evaluated by recording indicators such as body weight and disease activity index (DAI). Furthermore, 16S rDNA sequencing was employed to analyse changes in the gut microbiota, and alterations in serum inflammatory marker levels were detected to gain further insightthe anti-inflammatory mechanisms of 2′-FL.
Key Findings
1. 2′-FL alleviates colitis disease symptoms
Compared to the control group with DSS-induced colitis, mice treated with 2′-FL exhibited a lower proportion of weight loss attributable to disease and significantly reduced disease activity index (DAI) scores. This indicates that 2′-FL effectively mitigates intestinal inflammatory symptoms, with the most pronounced effect observed in the high-dose group.
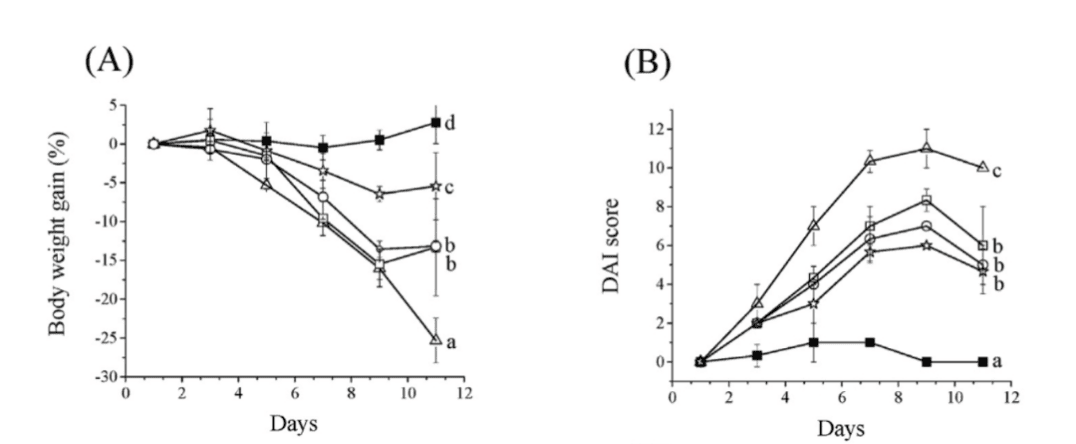
Figure 1: Effect of 2′-FL on colitis symptoms in a mouse model of 3% DSS-induced colitis
2. 2′-FL improves gut microbiota balance and metabolism
16S rDNA sequencing revealed that 2′-FL significantly restored the balance of gut microbiota disrupted by DSS-induced dysbiosis, increasing the proportion of beneficial bacteria such as Lactobacillus. Furthermore, 2′-FL promoted the production of beneficial bacterial metabolites known as short-chain fatty acids (SCFAs). These metabolites not only aid in maintaining intestinal barrier function but also exert anti-inflammatory effects.
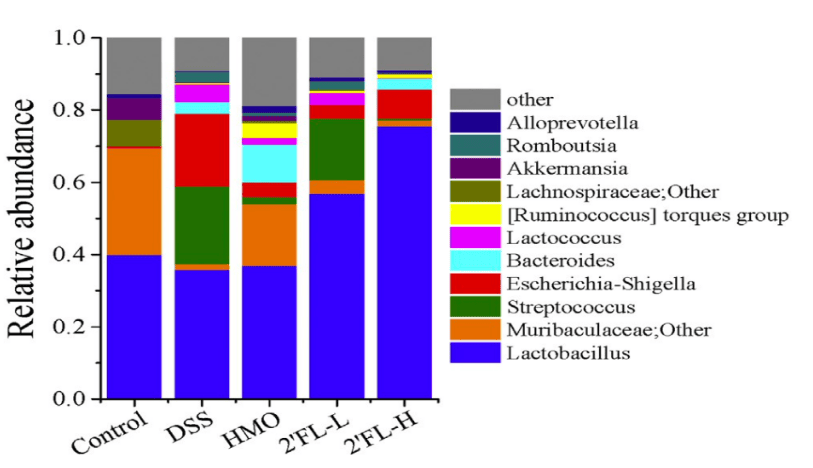
Figure 2: Comparison of gut microbial communities across different groups
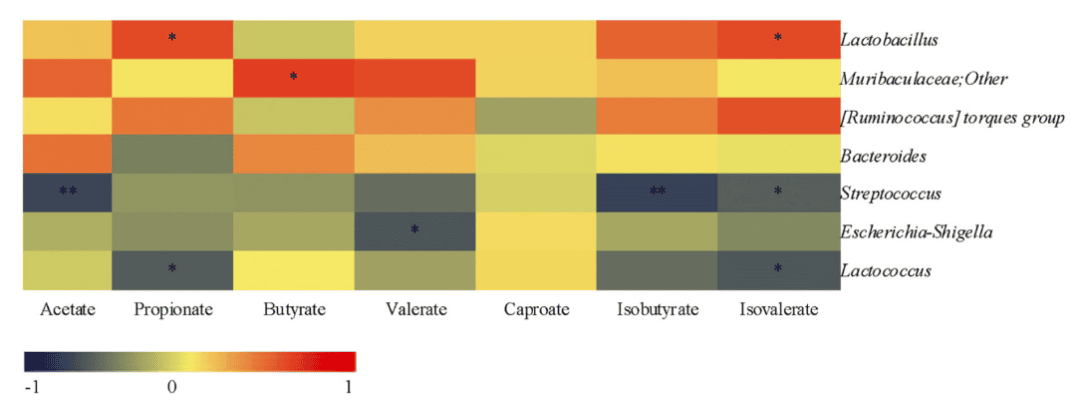
Figure 3: Heatmap of Spearman correlations between gut microbiota and SCFAs
3. 2′-FL Modulates Inflammatory Cytokine Levels
Serum analysis revealed that 2′-FL effectively reduced levels of the pro-inflammatory cytokines TNF-α and IL-6 in colitis-affected mice, while simultaneously increasing expression of the anti-inflammatory cytokine IL-10, thereby diminishing the inflammatory response.
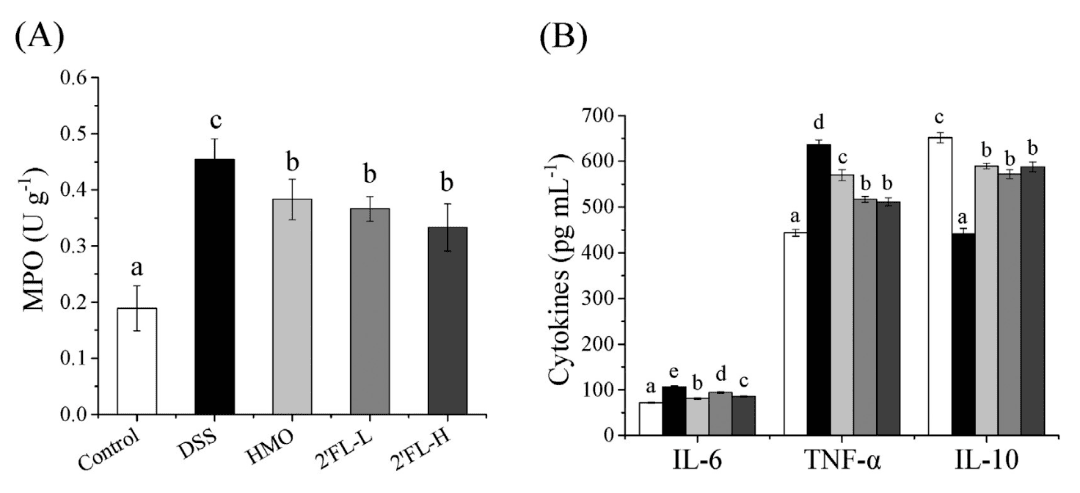
Figure 4: Effect of 2′-FL on Serum Levels of Inflammatory Markers
Discussion and Outlook
This study has demonstrated the beneficial effects of 2′-FL in alleviating inflammatory bowel disease (IBD). 2′-FL not only supports gut health by improving the gut microbiota and promoting the production of beneficial metabolites, but also significantly reduces intestinal inflammation by regulating immune responses through balancing pro-inflammatory and anti-inflammatory factors. Future research may further explore the clinical application potential of 2′-FL in IBD patients and investigate its role in other intestinal disorders and immune health management, particularly its potential in promoting gut health and immunomodulation.
Reference
[1] Li A, Ni W, Li Y, et al. Effect of 2′-fucosyllactose supplementation on intestinal flora in mice with intestinal inflammatory diseases[J]. International Dairy Journal, 2020, 110: 104797.
