
background
The intestinal barrier serves as a vital defense system for maintaining gut health and immune homeostasis, with its dysfunction closely associated with various chronic diseases. Human milk oligosaccharides (HMOs), particularly 2’-fucosyllactose (2'-FL), demonstrate extensive biological activities in regulating gut microbiota, anti-inflammatory immunity, and preserving intestinal barrier integrity. Previous studies have shown that HMOs not only act as a "carbon source" for probiotic growth but also enhance epithelial barrier function through specific mechanisms via their metabolites. This study investigates how 2'-FL (produced by Hongmo Biotechnology Co., Ltd.) alleviates LPS-induced intestinal barrier damage in vitro by modulating surface proteins of Bifidobacterium DNG6, providing insightsits underlying mechanisms.
Research design and methods
This study employed a Caco-2 intestinal epithelial cell monolayer in vitro barrier model to simulate LPS-induced intestinal inflammation. Bifidobacterium DNG6 was cultured under three distinct carbon sources: lactose (Lac), galactooligosaccharide (GOS), and 2 -fucosylated lactose (2'-FL). Surface proteins (labeled as Sp-L, Sp-G, and Sp-F) were extracted, and the LPS-treated Caco-2 cells were subjected to intervention. The effects were evaluated through cell viability, cytotoxicity, inflammatory factor expression, and tight junction protein levels.
Key findings
1. 2'-FL enhances cell viability and reduces cytotoxicity
Studies demonstrate that LPS treatment significantly reduces Caco-2 cell viability. Notably, Sp-F (2'-FL treatment group) achieves a cell survival rate exceeding 88% at 800–1000 μg/mL, outperforming both Sp-L and Sp-G. Furthermore, Sp-F (2'-FL treatment group) markedly decreases lactate dehydrogenase (LDH) release levels, a biomarker of cell membrane integrity, indicating its potent efficacy in mitigating membrane damage.
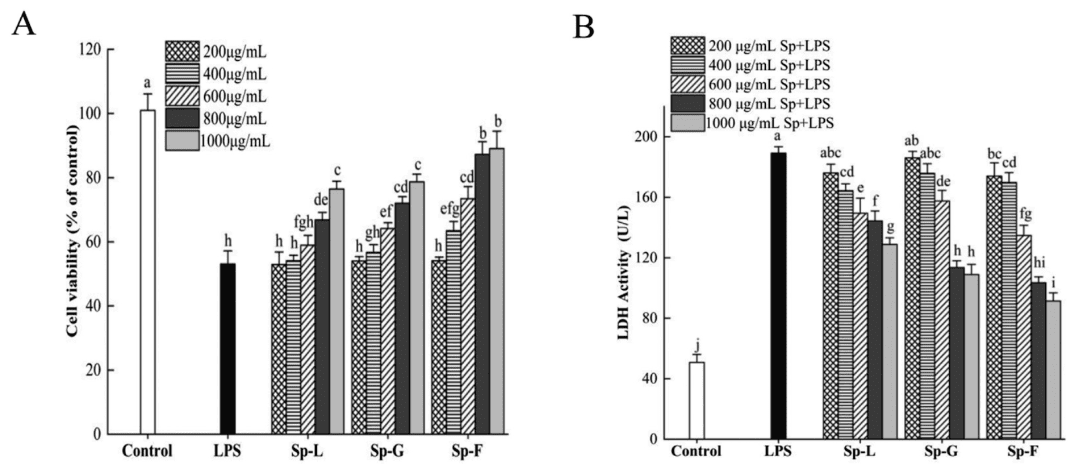
Figure 1: Effects of surface proteins on LPS-induced viability and LDH activity in Caco-2 cells
2. 2'-FL suppresses inflammatory responses and enhances immune regulation
LPS significantly induced the expression of pro-inflammatory factors TNF-α, IL-6, and IL-1β, while Sp-F (2'-FL treated group) significantly suppressed their secretion levels, reducing them by 53.46%,35.91%, and 37.05% respectively. Meanwhile, the anti-inflammatory factor IL-10 levels increased to 140.13% of the LPS group, indicating its excellent immune-regulating function.
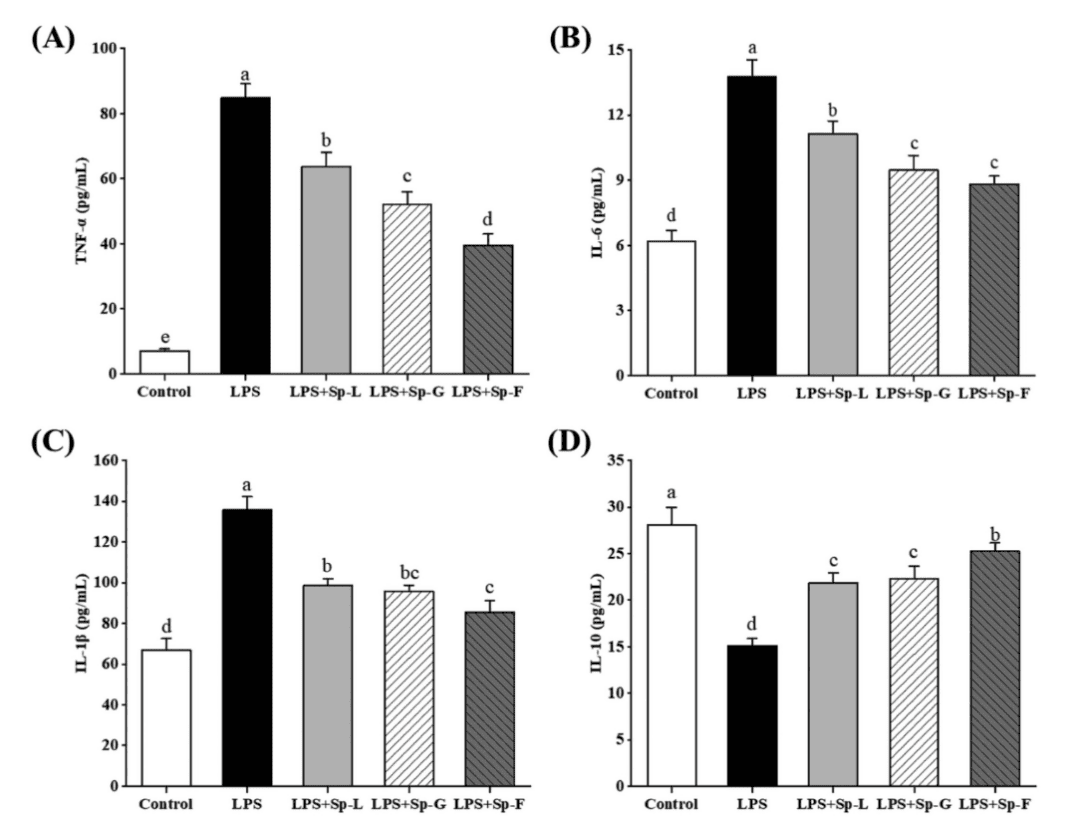
Figure 2: Effects of surface proteins on the expression of TNF-α, IL-6, IL-1β, and IL-10 in LPS-induced monolayer barrier of Caco-2 cells
3. 2'-FL repairs barrier structures and enhances cell junctions
In vitro experiments demonstrated that Sp-F (2'-FL treated group) significantly enhanced the mRNA expression (by 146.21%,268.53%, and 137.22% respectively) and protein levels of ZO-1, Claudin-1, and Occludin, which are tightly junction proteins in intestinal epithelial cells. These proteins form critical structures that maintain intestinal barrier integrity and prevent harmful substance permeation. The results indicated that Sp-F effectively repaired the intestinal epithelial barrier and improved intercellular junction integrity.
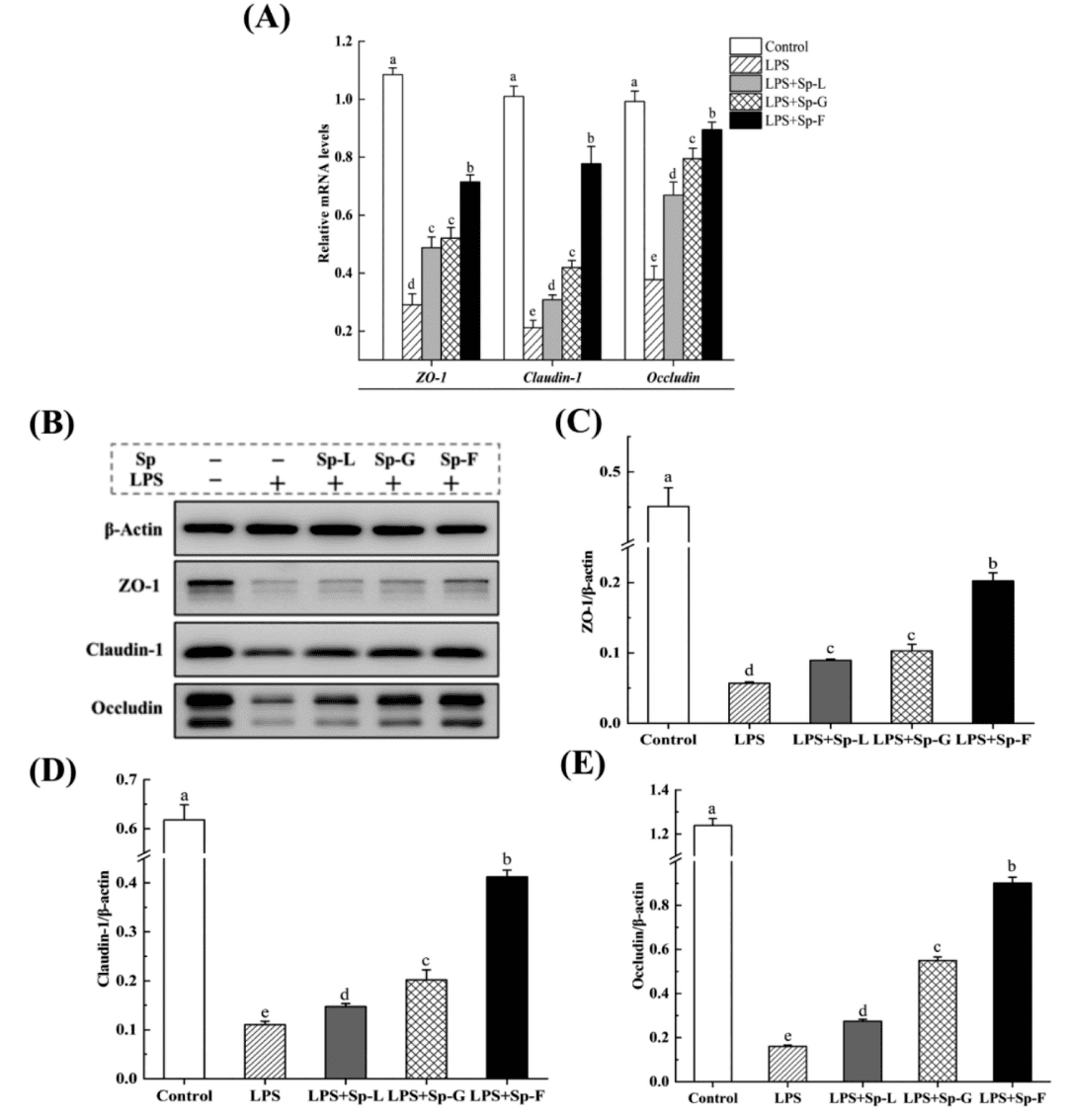
Figure 3: Surface protein effects on protein mRNA and protein expression
Discussion and outlook
This study reveals the mechanism of 2'-FL (produced by Hongmo Biotechnology Co., Ltd.) in maintaining intestinal barrier homeostasis from the perspective of "bacterial surface proteins": By promoting the expression of surface proteins in Bifidobacterium DNG6, it significantly alleviates LPS-induced inflammatory damage and enhances tight junction protein expression. This discovery not only expands the understanding of HMOs molecular mechanisms but also provides theoretical support for the functional application of HMO in infant formula. Future research could further integrate animal models and clinical studies to explore its potential applications in populations such as preterm infants and inflammatory bowel disease patients.
References
[1] Zhao, J., et al. "Surface Proteins of Bifidobacterium Bifidum DNG6 Growing in 2'-FL Alleviating LPS-Induced Intestinal Barrier Injury in vitro." Journal of Dairy Science (2024): S0022-0302.
